How to Find Lowest Osmotic Pressure
Calculate the osmotic pressure given the number of grams of sucrose dissolved in water. 1 psi 6894810-2 bar.

1 Relationship Between Concentration And Osmotic Pressure Of Solution Download Scientific Diagram
Osmotic pressure is a colligative substance property because it depends on the concentration of the solute but not its chemical nature.

. Π 0272 molL 0082 l atmmol K 310 K 69 atm. March 27 2022 Proteins in blood plasma are used to measure colloid osmotic pressure. As blood moves through the capillaries it filters into the tissue space delivering nutrients to the cells.
The protein does not dissociate in water. FeCl3 003 4 012. C molar concentration of solute R ideal gas constant T temperature in Kelvin.
PLEASE MARK ME AS A BRAINLIEST. 3 Which leads to this van t Hoff factor. Calculate the osmotic pressure of this solution.
Osmotic pressure icRT. Mol-1K-1 300 K π 4926 atm. C is the molar concentration of the solute in the solution.
1 Based on the 35 we have the following. So value of ic are. In the given problem CR and T are same for all three solutions.
The higher the concentration M or the temperature T of a solution the higher the osmotic pressure. HOPES U LIKE IT IT HELPS U. The Osmotic Pressure calculation example is given with a brief description below.
010 m K2SO4 releases 03 mol of ions. Follow edited Dec 10 2018 at 1418. 005 m BaCl2 releases 015 moles of ions.
Tonicity is the measure of this pressure. The osmotic pressure of the 1M salt solution is 4926 atmospheres at a temperature of 27 o C. 005 m CaSO4 releases 01 moles of ions.
Osmotic weight estimation might be utilized for the assurance of sub-atomic loads. Click to see full answer Similarly one may ask how does albumin maintain oncotic pressure. Where i Vant Hoff factor.
A decrease in oncotic pressure due to a low albumin level allows fluid to leak out from the interstitial spaces into the peritoneal cavity producing ascites. Question 18 5 points The osmotic pressure of 2000 mL of a 25C solution containing 020 g of a protein is 62 Torr. π 2 1 molL-1 00821 atmL.
R is the universal gas constant. This calculator converts automatically the pressure to bar with the following conversion factor. We can see from this equation that the amount of solute present in the solution will directly affect the osmotic pressure of the system.
Which of the following solutions has the lowest osmotic pressure. Katriena Knights Date. Albumin is essential for maintaining the oncotic pressure in the vascular system.
Where π is the osmotic pressure. A 16 x 101 b 16 x 103 Oc 30 x 103 d 16 x 104. Made by faculty at the University of Colorado Boulder Department of.
0015525 0015 1035. 2 The actual concentration of all species in solution H Ac-and HAc is this. LatexPi i M R Tlatex Here i is the van t Hoff factor M is the molarity of the solution R is the gas constant and T is the absolute temperature in Kelvin.
The equation for osmotic pressure is piiMRT. To find the osmotic pressure plug the values into the equation. Which of the following solutions has the lowest osmotic pressure.
I is the vant Hoff factor. 010 m KOH releases 02 mol ion. Hypertonicity is the nearness of an answer that makes cells shrivel.
Determining the van t Hoff factor. Finally find the osmotic pressure using Π MRT equation. Osmoregulation is the homeostasis system of a living being to arrive at balance in osmotic weight.
Therefore the osmotic pressure of the solution is. Osmotic Pressure Vocabulary. Osmotic pressure is the difference between blood in the capillaries and interstitial fluid between the cells according to Kimballs Biology Pages.
CaCl2 005 3 015. Since this interstitial fluid has a lower concentration of plasma than proteins and a relatively higher concentration of. APPLYING A OSMOTIC PRESSURE CALCULATOR.
Colloid osmotic pressure also referred to as oncotic pressure is a measurement of pressure exerted within the cardiovascular system by proteins found in blood plasmaThe special nature of these protein cells helps ensure that fluids pass in. The osmotic pressure II of an ideal solution can be approximated by the Morse equation. K2FeCN6 004 4 016.
010 m RbCl releases 02 moles of ions. Π iMRT Π 1 x 016 molL x 008206 LatmmolK x 298 K Π 39 atm Answer. The osmotic pressure of a potassium chloride solution at 300K is 50 atmospheres.
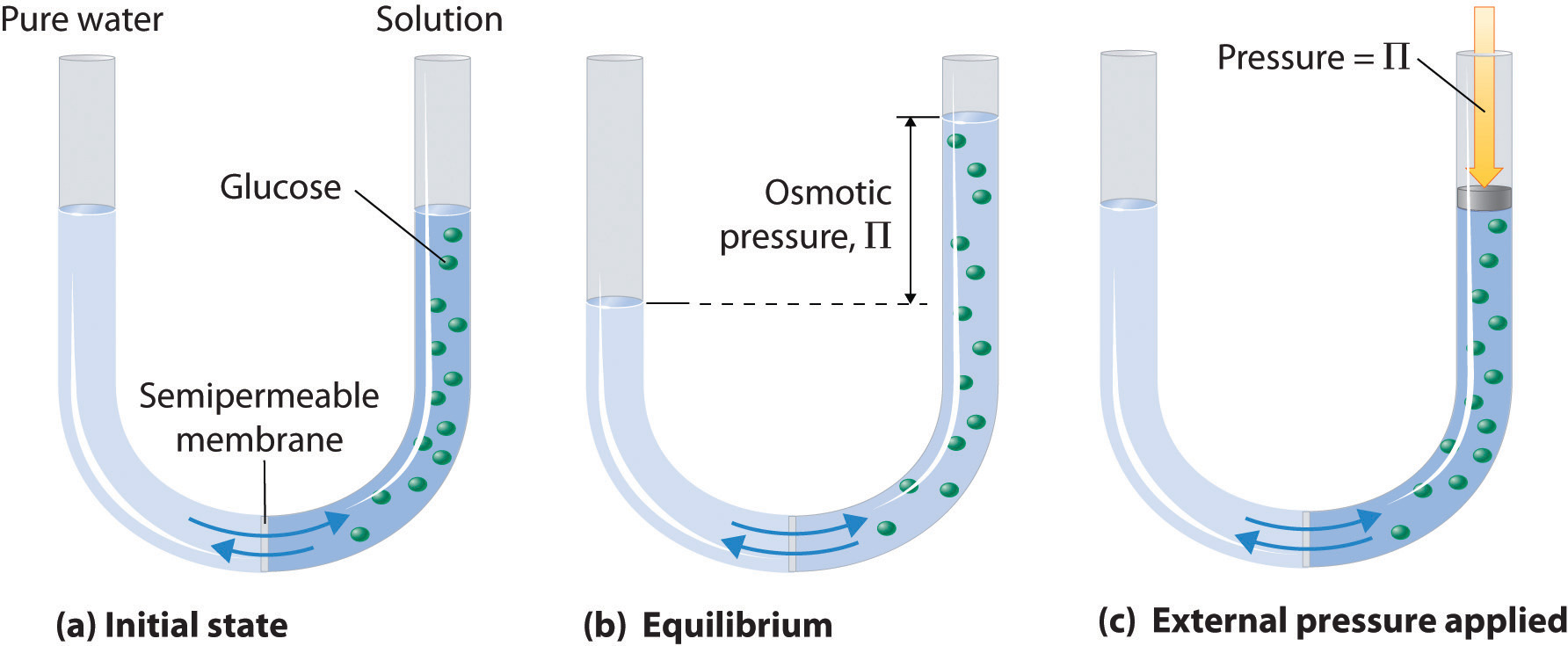
Osmotic pressure is the pressure of a solution against a semipermeable membrane to prevent water from flowing inward across the membrane. RT is same for all. NaCl 0010 2 0020 002.
010 M CaBr2 B. Chemistry questions and answers. CeMgCl2 has the highest osmotic pressure because it breaks up into three ions in water as opposed to all the others which only break up into two.
The obtained osmotic pressure with formula 2 is in psi pounds per square inch. Therefore NaCl shows the lowest osmotic pressure. 015 M Ba NO22.
In reality of course osmotic pressure is not a desire of water to move but rather an extension of the natural law that all. Glucose will have the lowest osmotic pressure because it has the lowest i value. Osmotic pressure is determined by solute concentration water will try harder to diffuse into an area with a high concentration of a solute such as a salt than into an area with a low concentration.
0000525 0000525 0015 - 0000525 0015525 M. The osmotic pressure of the sucrose solution is 39 atm. Osmotic pressure is directly related to the moles released.
Based on this information determine the molar mass of the protein in gmol. H Ac- 0015 0035 0000525 M. Osmotic weight is a significant factor influencing cells.
Osmotic pressure π iCRT. If you do not have a water analysis you can use the values given in. Thus the Vant Hoff factor equals 1.
A low serum albumin indicates. To calculate osmotic pressure use the following formula. T is the temperature.
If the concentration of solutes on both sides of the membrane is equal then there is no tendency for water to move across the membrane and no osmotic pressure. Calculating the osmotic pressure formula chemistry is done using π iMRT.

Cells 7 Osmotic Pressure Physics And Mathematics Cell Membrane
What Is The Osmotic Pressure Of A 0 050 M Solution Of Alcl 3 In Water That Is At 0 00 C Socratic

Osmotic Pressure Physiology Youtube

Osmotic Pressure Definition Formula Video Lesson Transcript Study Com

Osmotic Pressure As A Function Of Solution Concentration At 25 C For Download Scientific Diagram

Among The Following The Solution Which Shows The Lowest Osmotic Pressure Is A 0 14 M Nacl B Youtube
1 Osmotic Pressure Of Single Salt Solutions As Related To The Download Scientific Diagram

Osmotic Pressure Definition Formula Video Lesson Transcript Study Com

The Osmotic Pressure Of Equimolar Solutions Of Bacl 2 Nacl And Glucose Follow The Order Youtube
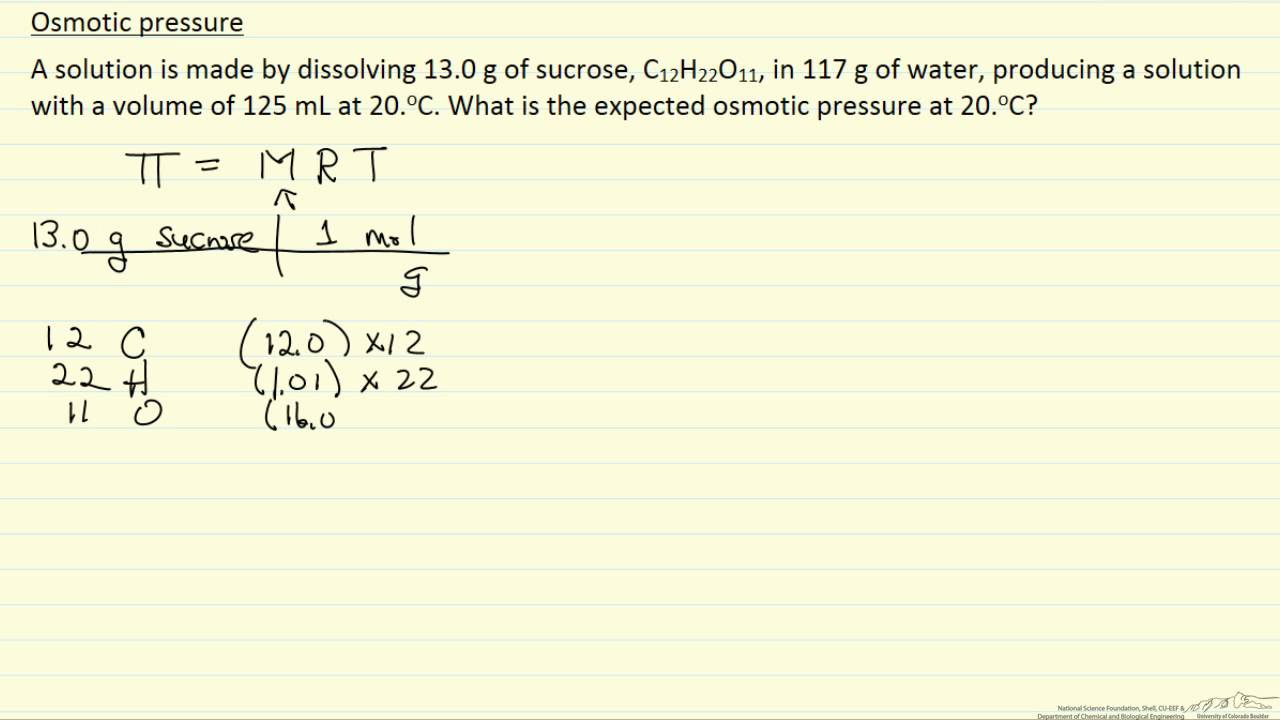
Osmotic Pressure Example Youtube

Osmotic Pressure Of Salt Solutions Download Scientific Diagram

Osmotic Pressure Of Nacl Solution At Different Temperatures And Download Scientific Diagram

Calculate Osmotic Pressure Of A Solutions Having 0 1m Nacl 0 2m Na 2 So 4 And 0 5m Youtube

How Many Millilitres Of 0 1 M H2so4 Must Be Added To 50 Ml Of 0 1 M Naoh To Give A Solution That Has A Concentration Of 0 05 M In H2so4
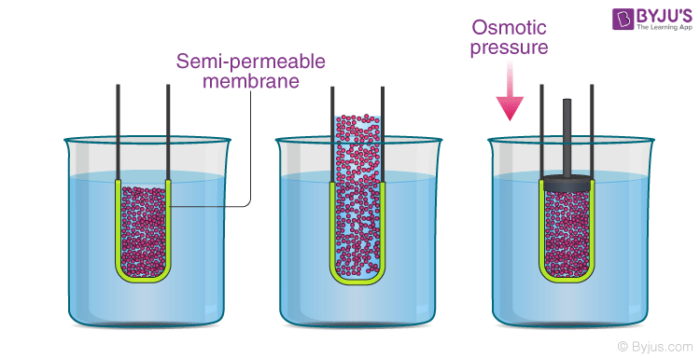
Osmotic Pressure Definition Formula Examples Solved Exercises

Comparisons Of Predicted A Osmotic Pressure P Os And B Osmotic Download Scientific Diagram
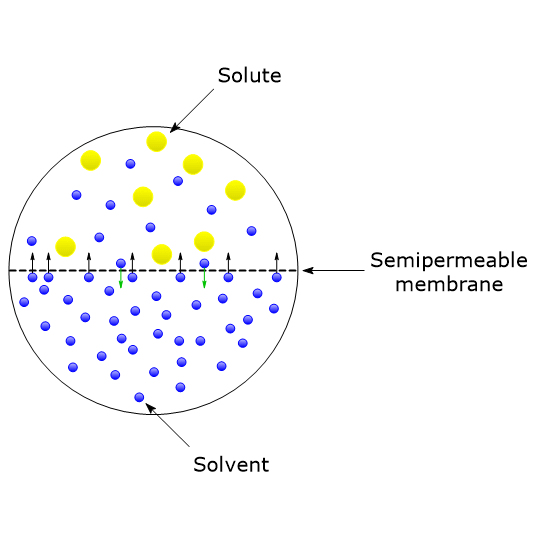
Osmotic Pressure Definition Formula And Importance

Osmotic Pressure And Concentration G L Of Draw Solution Download Scientific Diagram
Comments
Post a Comment